myCSG © |Area: SDTM| Concept: IE : Inclusion/Exclusion Criteria Not Met | Lesson: L101 : Inclusion/Exclusion Criteria Not Met |
Background |
|
What type of information is typically collected or presented? |
|
Other notes |
|
In this lesson,
We will see a basic example on how to create IE domain
ELIGCRIT : Eligibility Criteria
| Question | Response |
|---|---|
| Date of assessment [IEDAT] |
|
| Inclusion Criteria 1 [ICRIT01] |
Yes? No |
| Inclusion Criteria 2 [ICRIT02] |
Yes? No |
| Exclusion Criteria 1 [ECRIT01] |
Yes? No |
| SUPP | VARIABLE_NAME | VARIABLE_LABEL | TYPE | GENERAL_COMMENT | FORM_ELIGCRIT |
|---|---|---|---|---|---|
| _ | STUDYID | Study Identifier | Char | Set to 'statschoice' | |
| _ | DOMAIN | Domain Abbreviation | Char | Set to 'IE' | |
| _ | USUBJID | Unique Subject Identifier | Char | Concatenate eligcrit.PROJECT and eligcrit.SUBJECT with a hyphen in between. | |
| _ | IESEQ | Sequence Number | Num | Sort the records by USUBJID, IECAT, IETESTCD, IEDTC, IESPID and assign as 1 on the first record of each subject and increment by 1 for each subsequent record for that subject. | 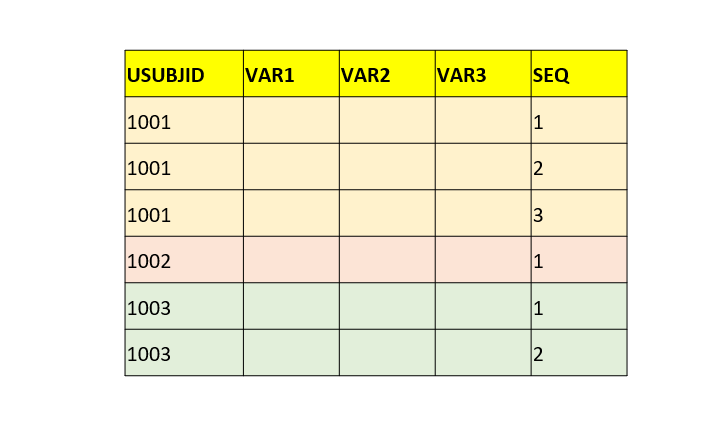 |
| _ | IESPID | Sponsor-Defined Identifier | Char | Populate using eligcrit.RECORDPOSITION in three digit format padding with leading 0's where applicable. | |
| _ | IETESTCD | Inclusion/Exclusion Criterion Short Name | Char | 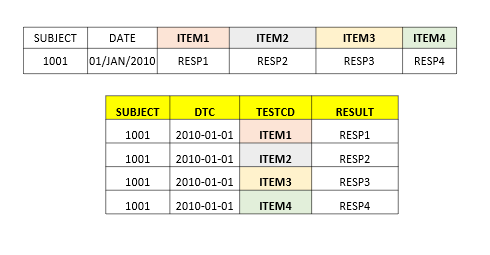 |
Create a record for each failed inclusion criteria (result=No) and each met exclusion criteria (result=Yes). Populate as 'INCL01' when creating a record using eligcrit.ICRIT01. Populate as 'INCL02' when creating a record using eligcrit.ICRIT02. Populate as 'EXCL01' when creating a record using eligcrit.ECRIT01. |
| _ | IETEST | Inclusion/Exclusion Criterion | Char | Populate as 'Inclusion criteria 1' when SDTM.IE.IETESTCD=INCL01. Populate as 'Inclusion criteria 2' when SDTM.IE.IETESTCD=INCL02. Populate as 'Exclusion criteria 1' when SDTM.IE.IETESTCD=EXCL01. |
|
| _ | IECAT | Inclusion/Exclusion Category | Char | Populate as 'INCLUSION' for the records with SDTM.IE.IETESTCD begins with INCL, and populate as 'EXCLUSION' for the records with SDTM.IE.IETESTCD begins with EXCL. | |
| _ | IEORRES | I/E Criterion Original Result | Char | Populate as 'N' when creating a record for INCLxx. Populate as 'Y' when creating a record for EXCLxx. |
|
| _ | IESTRESC | I/E Criterion Result in Std Format | Char | Populate using SDTM.IE.IEORRES | |
| _ | VISITNUM | Visit Number | Num | Populate using eligcrit.FOLDERSEQ | |
| _ | VISIT | Visit Name | Char | Populate using eligcrit.FOLDERNAME | |
| _ | EPOCH | Epoch | Char | Populate using SDTM.SE.EPOCH by comparing IEDTC with SESTDTC and SEENDTC of each element of the subject. Populate as 'TREATMENT' when the record falls on the boundary of SCREENING and TREATMENT elements. Populate as 'TREATMENT' when the record falls on the boundary of TREATMENT and FOLLOW-UP. | 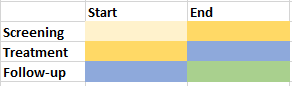 |
| _ | IEDTC | Date/Time of Collection | Char | Populate using eligcrit.IEDAT in ISO 8601 format. | |
| _ | IEDY | Study Day of Collection | Num | If the SDTM.IE.IEDTC is on or after reference start date then SDTM.IE.IEDTC-SDTM.DM.RFSTDTC +1. Else if date is before reference start date then assign as SDTM.IE.IEDTC-SDTM.DM.RFSTDTC. | 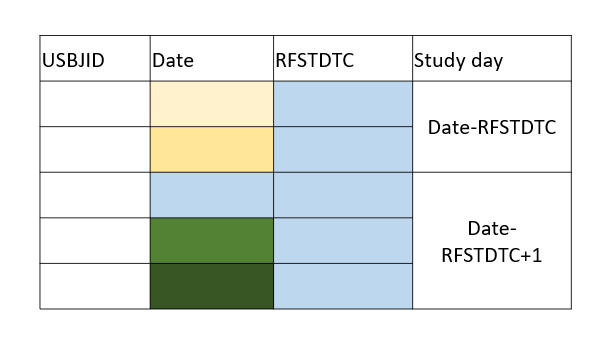 |
Complete SAS code to generate the output is available for registered users!
Already registered! Login
Not registered, you can signup here! Signup
Dont want to register?
You can directly purchase this lesson (code+input data) here
Already registered! Login
Not registered, you can signup here! Signup
Dont want to register?
You can directly purchase this lesson (code+input data) here
You can purchase this lesson (code+input data) here
If you are looking to purchase subscription for full access to data and programs for all lessons (TASKS+SDTM+ADaM+TFLs), you can send us a message on +91-7330--77--66--49-- on Whatsapp.
R data is available only for R subscribers.
You can contact us on +91-7330--77--66---49 for purchasing subscription to R programs and data
You can contact us on +91-7330--77--66---49 for purchasing subscription to R programs and data
R codes are available only for R subscribers.
You can contact us on +91-7330--77--66---49 for purchasing subscription to R programs and data
You can contact us on +91-7330--77--66---49 for purchasing subscription to R programs and data